SMS for Lab Result Notifications, What's Allowed and What's Not
- Praveen Suddala

- 4 hours ago
- 13 min read

Patients want their lab results the moment they are ready. They check their phones an average of 144 times a day according to data from Reviews.org, but they log into a clinical patient portal less than once a month according to multiple healthcare engagement studies. That gap is why so many laboratories, diagnostic imaging centers, and physician offices are turning to SMS for lab result notifications.
The trouble is that text messaging sits at the intersection of three overlapping rule sets. HIPAA governs the privacy and security of protected health information. The CLIA Final Rule of 2014 gives patients a direct legal right to their own results. The 21st Century Cures Act information blocking rules require that those results be shared without unreasonable delay. Get the SMS workflow right and you cut callbacks, free up phone lines, and give patients exactly what they want. Get it wrong and you are looking at six and seven figure HIPAA settlements, CLIA citations, and information blocking complaints.
This guide walks through exactly what labs can and cannot do over SMS in 2026, what consent really requires, the texts that pass a compliance audit, and how a HIPAA-ready platform like Falkon SMS lets you send the actual PDF lab report by text without exposing PHI.
Why Labs Are Moving to SMS in the First Place
The numbers behind the shift to texting are hard to ignore. SMS open rates sit between 95 and 98 percent within three minutes of delivery according to multiple carrier studies. Email opens average 21 percent. Patient portal logins for routine results are even lower. For a lab whose entire downstream workflow depends on the patient seeing the result, texting is not a marketing channel, it is the most reliable patient touchpoint available.
The operational case is just as strong. Every result that the patient does not see by themselves becomes a phone call to the ordering provider, a callback from a medical assistant, or a follow up appointment that did not need to happen. A 250 patient per day reference lab can easily save 30 to 50 staff hours a week by switching routine result alerts to SMS.
That demand is exactly why HIPAA, CLIA, and ONC have all weighed in on what is and is not allowed.
The Short Answer on Whether You Can Text Lab Results
Yes, you can use SMS for lab result notifications, but only if you do all of the following.
You obtain documented patient consent to receive results by text, including an acknowledgement that SMS is not a fully secure channel. You restrict the content of the message itself so that no specific protected health information is exposed in the SMS body unless the patient has explicitly chosen that delivery method in writing. You use a texting platform that signs a Business Associate Agreement, encrypts data in transit and at rest, and produces a tamper proof audit log. And you give patients a way to access the full report through a secure channel such as a portal link or a password protected document delivered via secure SMS.
Standard carrier to carrier SMS does not, on its own, meet HIPAA's Security Rule technical safeguards. That is the single most misunderstood point in the entire industry, and it is where most violations begin.
What HIPAA Actually Says About SMS for Lab Results
HIPAA does not mention SMS by name. Instead it sets requirements for any electronic transmission of PHI, and SMS has to be evaluated against those requirements.
The Privacy Rule and the Right to Confidential Communications
Under 45 CFR 164.522, patients have the right to request that a covered entity communicate with them by alternative means, including by text message. If a patient submits that request and you decline without a reasonable basis, you are out of compliance with the Privacy Rule. HHS has confirmed in multiple guidance updates that this includes SMS where the patient has been informed of the risks and still wants the result delivered that way.
In other words, the Privacy Rule actively supports texting patients when the patient asks for it. The Security Rule is what controls how you do it.
The Security Rule Technical Safeguards That Apply to Texting
The HIPAA Security Rule requires five technical safeguards that any electronic transmission of PHI must satisfy. These are access controls, audit controls, integrity controls, person or entity authentication, and transmission security. SMS only satisfies these when it is sent through a HIPAA-ready platform that adds the missing layers.
The encryption standard most commonly accepted is AES-256 at rest and TLS 1.2 or higher in transit. Audit logs need to capture who sent the message, when, to whom, and what action followed. Access to the sending console must be tied to unique user credentials with role based permissions, not a shared mailbox.
Why Carrier SMS Is Not HIPAA Compliant by Itself
A standard SMS travels through carrier infrastructure that the lab does not control. Copies can sit on carrier servers indefinitely. Messages can be forwarded by the recipient. There is no signed BAA between the lab and the carrier. There is no audit trail that ties a specific employee to a specific message. For all of those reasons, OCR has consistently treated raw SMS containing PHI as a compliance failure.
This is why the rule of thumb across the industry is simple. The fact that a result is ready can be communicated by SMS. The result itself, in any detail that identifies the test or the finding, should be delivered through the secure layer that sits on top of SMS.
The CLIA Final Rule and Direct Patient Access
In 2014 CMS finalized changes to the Clinical Laboratory Improvement Amendments that gave patients a federal right to receive their completed lab test reports directly from the lab on request. This pre-empted state laws in roughly two thirds of states that previously required a clinician to be the gatekeeper for results.
For SMS workflows the implication is direct. A lab cannot use a state law as a reason to refuse to text a patient their results, because the federal rule controls. At the same time the lab is responsible for verifying the identity of the patient before releasing results. That is why every compliant SMS lab notification flow starts with patient verification at the point of consent collection, not at the moment the result drops.
The 21st Century Cures Act and Information Blocking
The Cures Act information blocking rules took full effect for laboratories and other actors on October 6, 2022. Under those rules a lab cannot impose unreasonable delays on releasing results to patients, including waiting for a clinician review window that no longer has a clinical justification.
Most labs respond to this by releasing results to the portal as soon as they are signed out. SMS notifications fit naturally with that workflow, since a text alert that says the report is available in the portal removes the patient side delay between sign out and patient awareness. The information blocking rules are not a green light to text raw values, but they are a strong reason to use SMS as the alert layer that gets patients to their results faster.
What You Are Allowed to Text
Below is the operating list most compliance teams converge on for SMS for lab result notifications. The unifying principle is that the SMS body should be informative enough to drive action, but stripped of any specific clinical finding that could disclose PHI to an unauthorized viewer.
Result ready alerts, using only a first name and the lab's name.
Secure links to a portal or authenticated viewer where the patient can see the full report.
Follow up appointment requests, without specifying the medical reason.
Specimen received confirmations letting patients know processing is underway.
Action required nudges, such as an unsigned consent or an outstanding balance blocking release.
Identity verification prompts, such as a request to reply with a four digit code from a visit slip before release.
Password protected PDF lab reports, but only when delivered through a HIPAA-ready secure file sharing layer (more on that below).
What You Are Not Allowed to Text
The list of what does not belong in a raw SMS body is just as important. Anything in this list, sent as plain SMS without the secure layer on top, is a HIPAA violation waiting to be cited.
Sensitive test names that imply a condition, for example HIV, hepatitis, pregnancy, drug screen, BRCA, or sexually transmitted infection panels.
Numerical results or interpretation language, such as positive, negative, abnormal, or critical.
Diagnostic codes, including ICD-10 codes that effectively describe the condition.
Patient's full name plus a lab name when the lab itself is identifying, for example a fertility clinic or a behavioral health lab.
Provider names tied to a sensitive specialty in the same message.
Photos, scanned images, or PDFs sent as a regular MMS attachment without encryption.
Direct identifiers, such as a medical record number, date of birth, or social security number.
A useful test is the elevator test. If the SMS were read out loud in an elevator full of strangers, would anyone learn something private about the patient. If the answer is yes, it does not belong in the body of the text.
The Patient Consent Workflow That Keeps You Safe
Consent is the single most audited element of any SMS lab program, and it is also the most common point of failure. A defensible consent flow has five elements.
First, written consent that the patient wants to receive lab notifications by SMS. Second, an explicit acknowledgement that SMS is not a fully secure channel and that the patient understands the residual risk. Third, an opt out path the patient can use at any time, typically by replying STOP. Fourth, a clear scope, which includes whether the patient consents only to alerts, or also consents to receiving the full report through a secure link. Fifth, a record of when consent was given, by whom it was collected, and any subsequent changes.
The consent record needs to live somewhere the lab can produce in seconds during an audit. A platform that ties consent to the contact record, with a timestamp and the user who collected it, removes the manual chase that catches most labs out.
Lab Result Text Templates That Pass Compliance Review

Below are five SMS templates that have been used by working labs and reviewed by HIPAA counsel. Adjust the lab name, brand voice, and reply codes to your environment, and remove any element that does not match your own consent scope.
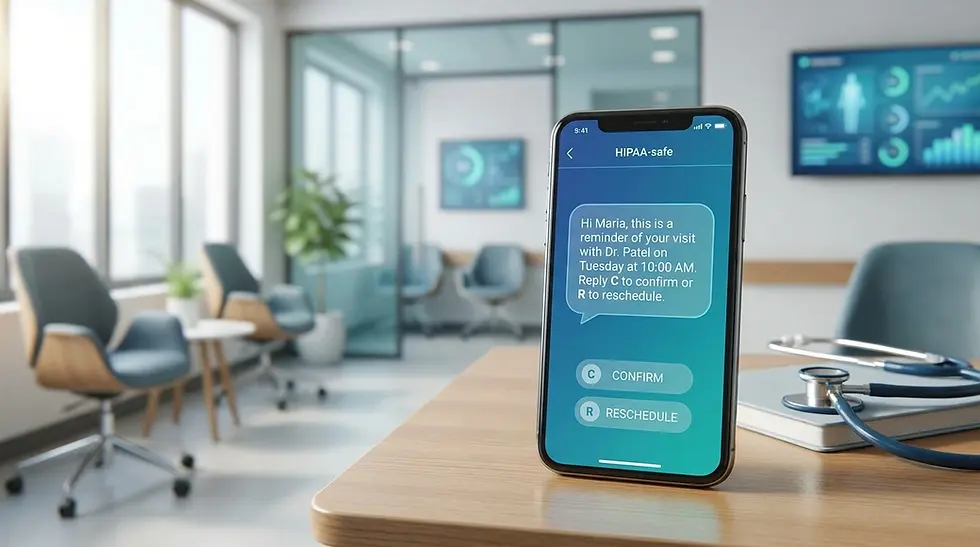
Template 1: Routine Results Ready
Hi Sarah, your recent lab work is back. View your full report securely here, [secure link]. Reply STOP to opt out. Northside Lab.
Template 2: Follow Up Required
Hi Mark, your results are ready and your provider has asked to discuss them. Please call 555 0142 or book online here, [link]. Reply STOP to opt out.
Template 3: Specimen Received
Hi Priya, we received your specimen on May 4 and processing is underway. We will text again when your report is available. Reply STOP to opt out.
Template 4: Action Required Before Release
Hi Daniel, your report is complete but we need a signed release on file before we can share it. Sign in 60 seconds here, [link]. Reply STOP to opt out.
Template 5: Identity Verification Before Release
Hi Lara, to release your report we need to confirm your identity. Reply with the four digit code on your visit slip. Reply STOP to opt out.
Notice what these templates avoid. No test name. No values. No sensitive specialty branding. No diagnostic language. The compliant action sits behind a secure link, not inside the SMS body.
How to Send the Full PDF Lab Report by Text Safely

There is no way to attach a PDF lab report to a regular MMS and stay compliant. The carrier infrastructure does not encrypt the attachment, the recipient device may back it up to an unencrypted cloud, and there is no audit log of who opened it.
The compliant pattern is a secure file sharing layer that lives inside the texting platform. The lab uploads the signed out PDF, the platform generates a one time access link with optional PIN protection and an expiration window, and the SMS sent to the patient contains only the link and a friendly note. When the patient taps the link, they authenticate inside the secure environment and view or download the report. Every open is logged to the audit trail.
This is exactly the workflow that Falkon SMS is built for. The platform is HIPAA ready with a signed Business Associate Agreement included in the Pro plan, every message and attachment is encrypted in transit and at rest, and Secure File Sharing lets a lab attach a PDF lab report to a text message via an expiring secure link rather than an unprotected attachment. Patients tap the link, optionally enter a PIN through Falkon's Secure Chat layer, and read the report inside an encrypted session with no app download. The lab gets the convenience of SMS, the patient gets the actual document, and the audit log captures the whole flow.
For a clinical lab that wants to text PDF lab reports without standing up a portal of its own, that combination of compliant SMS plus secure document delivery is the cleanest way to meet HIPAA, CLIA, and Cures Act expectations in one workflow.
Common Mistakes Labs Make With SMS Notifications
Five mistakes show up over and over again in HIPAA enforcement actions and CMS lab inspections, and almost all of them are easy to avoid once you know to look.
The first is collecting consent verbally and never writing it down. Without a timestamped record, the lab has no defense if the patient later complains. The second is using a personal cell phone or a generic SMS marketing tool that has no signed BAA. The third is putting the test name in the SMS body because the patient asked for it once, and continuing to do so for every patient. The fourth is forgetting to suppress identifiers in the SMS preview that shows on a locked phone screen, which can be visible to anyone holding the device. The fifth is not training new staff on what is and is not allowed in the SMS body, which is how a single rogue message becomes an OCR investigation.
A platform that enforces guardrails at the message level, rather than relying on the staff member to remember the rules, removes most of these failure modes.
A Compliance Checklist Before You Send Your First Lab Text
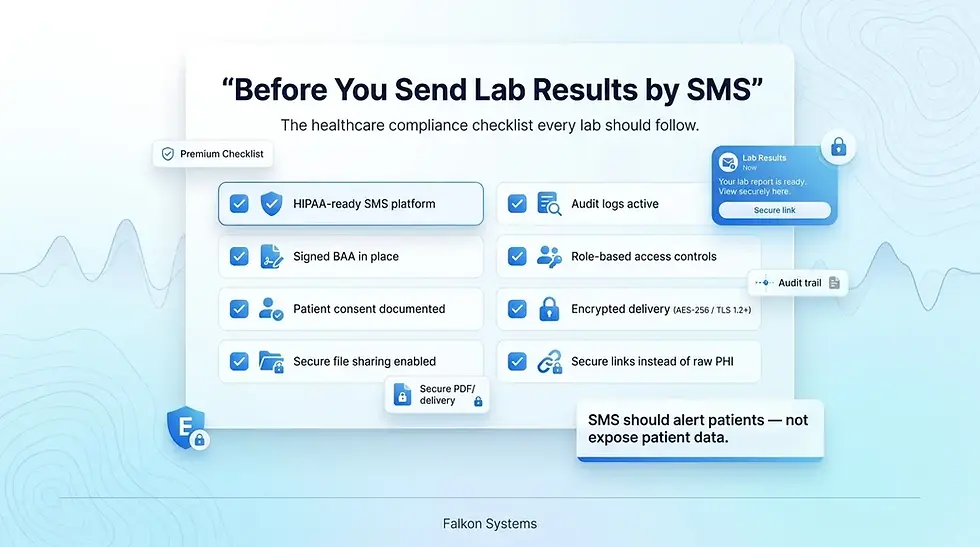
Use this checklist as a final pre flight before turning on SMS for lab result notifications. If any item is unchecked, fix it before the first message goes out.
A signed Business Associate Agreement with the SMS platform is in place. The platform encrypts data in transit using TLS 1.2 or higher and at rest using AES-256 or equivalent. Patient consent is collected in writing, scoped to lab notifications, and stored against the contact record. An opt out path is enabled and tested. SMS templates have been reviewed by privacy counsel and do not contain test names, values, or sensitive identifiers. A secure file sharing layer is configured for any case where the full report needs to be sent. Role based access controls are set so only authorized staff can send result notifications. An audit log is in place and exportable. Staff training on permitted and prohibited content is documented. A breach response plan covers SMS specific scenarios such as a wrong number, a forwarded message, or a compromised staff account.
Once those ten items are green, you are in the position most labs never reach, ready to text confidently and prove it.
Frequently Asked Questions

Can I text a patient their actual lab values
Only if the patient has given written, scoped consent that explicitly covers receiving values by SMS, and only through a HIPAA-ready platform that encrypts the message in transit and at rest. Even then, the safer pattern is to text an alert and deliver the values through a secure link or PIN protected report.
Is it a HIPAA violation to text a patient that their HIV test is ready
Yes, in almost every case. Naming a sensitive test in the SMS body discloses PHI to anyone who can see the screen, including a partner, a coworker, or a thief. Use a generic alert and require authentication for the actual result.
Do I need a signed BAA with my SMS provider
Yes. If the platform transmits, stores, or processes any PHI on your behalf, it is a Business Associate under HIPAA and must sign a BAA. Falkon SMS, for example, includes a signed BAA in its Pro plan.
Does the patient have a federal right to receive results by text
The patient has a right to request communications by an alternative means under the HIPAA Privacy Rule, and a right to receive their completed lab reports directly from the lab under the CLIA Final Rule. Together these create a strong case for honoring SMS requests as long as the lab can do so securely.
How fast does the Cures Act expect labs to release results
The Cures Act information blocking rule does not set a fixed time, but it prohibits unreasonable delays. Most labs aim to release to the patient at the same time the result is signed out, which is also the moment the SMS alert can fire automatically.
Can I text a PDF of the lab report
Not as a regular MMS attachment. You can text a secure link that points to a PDF inside an encrypted, expiring environment, which is how platforms like Falkon SMS handle document delivery without exposing PHI.
What happens if a patient replies with sensitive details
Your platform's audit trail and access controls protect the inbound message in the same way as outbound. Train staff to acknowledge the message in a non-clinical way, route the patient to a secure channel for any clinical discussion, and never forward sensitive content to personal email or messaging apps.
Are there state laws stricter than HIPAA on lab texts
A handful of states impose stricter rules on certain test categories, particularly HIV, mental health, and substance use disorder records covered under 42 CFR Part 2. Always check the strictest applicable rule. The federal CLIA right to access does not pre-empt narrower state confidentiality rules for these specific test types.
Final Thoughts
SMS for lab result notifications is one of the highest leverage workflow upgrades a lab can make in 2026. Patients see the alert in seconds. Phone volume drops. Information blocking risk goes down because patients learn about results faster. The catch is that SMS only works inside a compliance frame, with documented consent, encrypted infrastructure, a signed BAA, and a secure layer for the actual report.
Labs that get the frame right, and use a platform built for regulated industries, get all of the upside without any of the risk. The text becomes the alert, the secure link becomes the report, and the audit log becomes the proof.
If you are evaluating a platform to do this end to end, look for HIPAA readiness with a signed BAA, encryption in transit and at rest, secure file sharing for PDF lab reports, role based access, an immutable audit trail, and the ability to text from your existing lab phone number so patients recognize who is reaching out. Falkon SMS was built for exactly that profile, and it is one of the few platforms that combines all of those capabilities in a single workflow.
Send fewer phone calls. Send more compliant texts. Your patients, your lab, and your compliance officer will all thank you.